
Case Study: CTMS Implementation at CHUV Lausanne University Hospital
The Clinical Trial Unit within the CHUV Pharmacy required a system to efficiently track information on its growing portfolio of clinical studies. To address this, a custom Clinical Trial Management System (CTMS) developed at the CRC Lausanne was implemented. The software, built in Microsoft Access, enables simple data entry via a graphical user interface, activity tracking, and reporting.

Case Study: Redesigning an Intra-EU Clinical Supply Flow in SAP
Clinical trial supply chains are unforgiving. The moment a process is unclear, undocumented, or too manual to execute reliably under pressure, the risk lands in one place: delayed IMP delivery to patients.
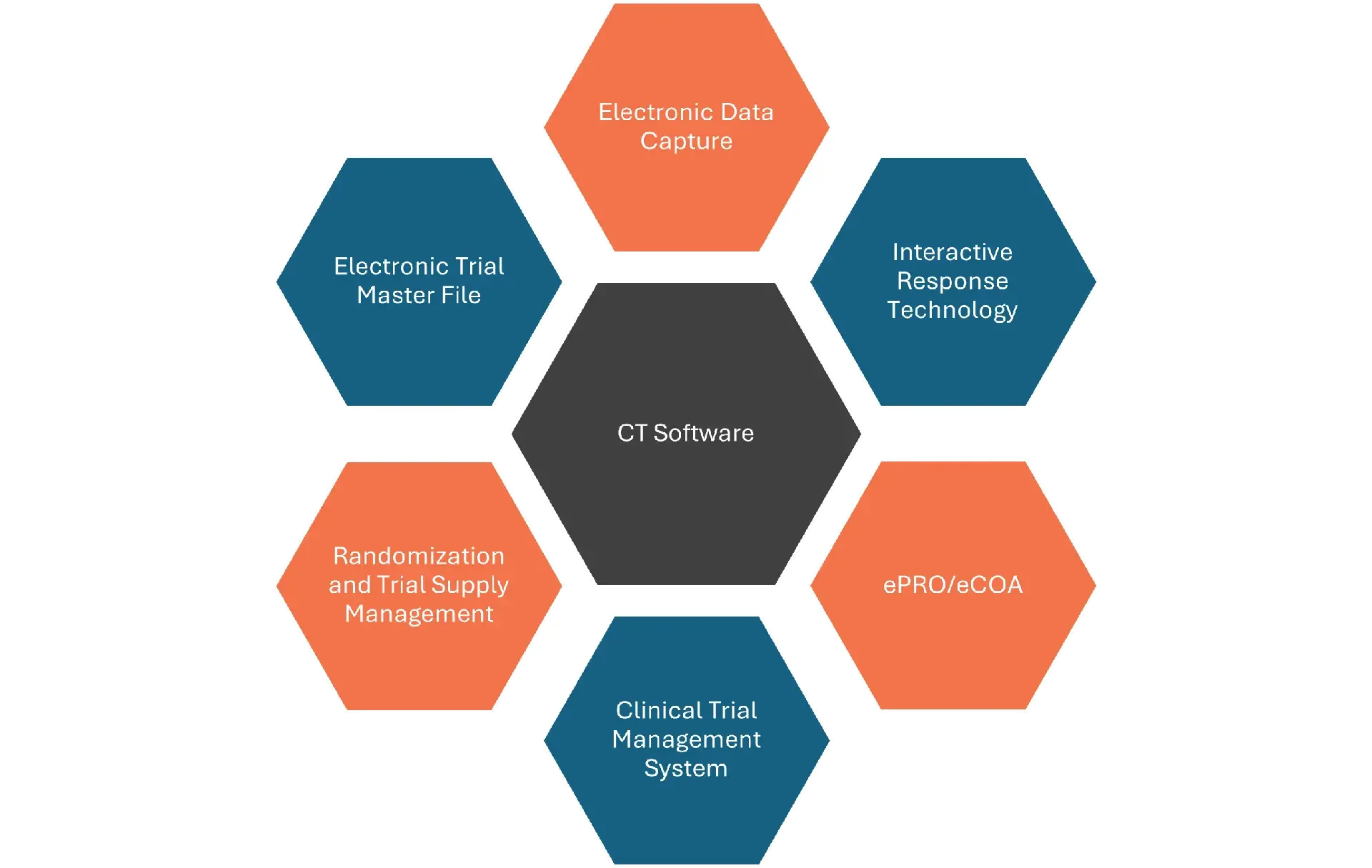
Where SAP ICSM Fits in the Clinical Trial Technology Landscape
Clinical trials run on software. The question is rarely whether a system is needed, it's which system does what, where the boundaries sit, and how the pieces connect.
What 21 CFR Part 11, Annex 11, and GAMP 5 Actually Mean for an SAP ICSM Implementation
Every SAP implementation in a pharmaceutical environment comes with a compliance layer. Everyone knows this. What is less often discussed is what that compliance layer actually requires in practice, and where it creates real implementation decisions rather than just documentation exercises.
