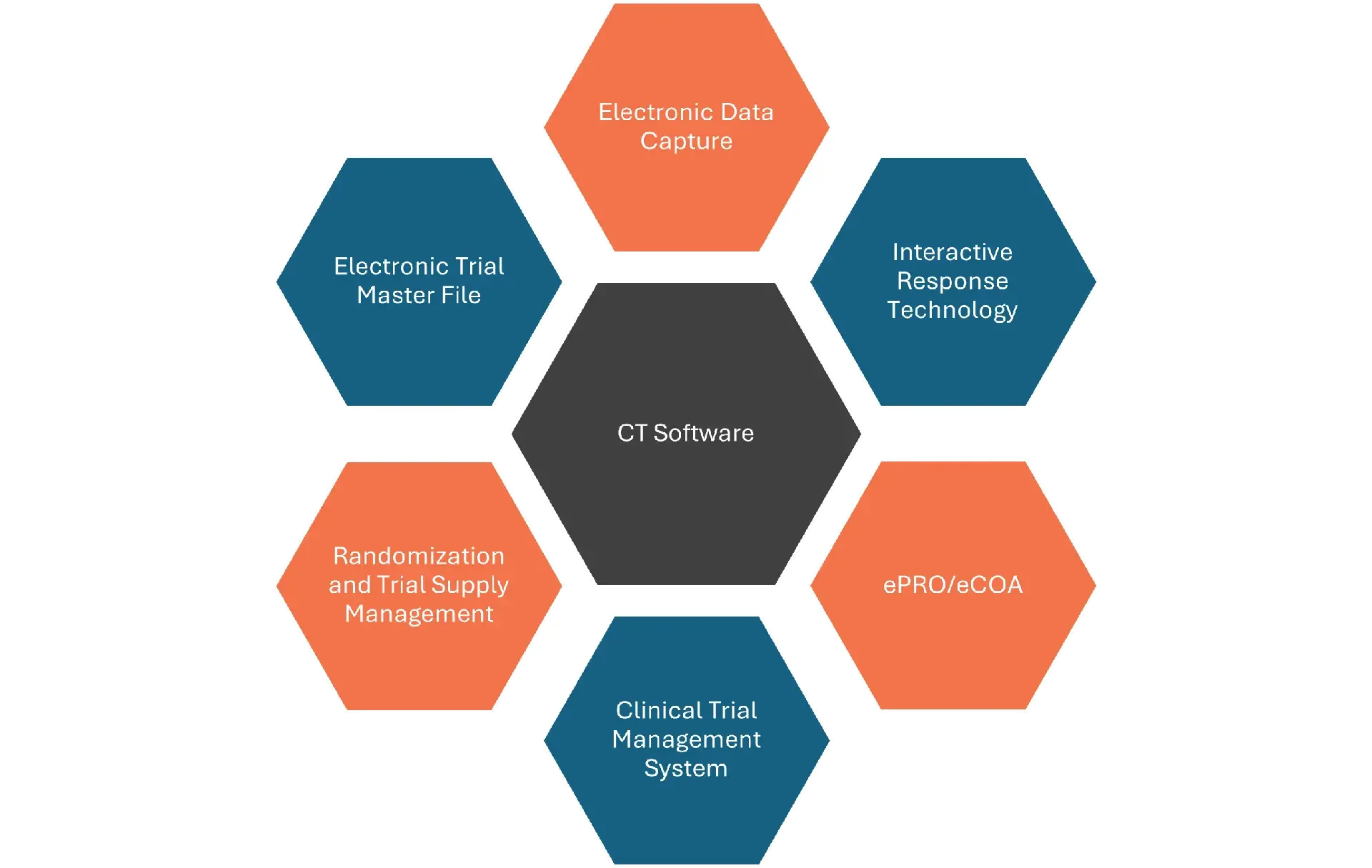
Where SAP ICSM Fits in the Clinical Trial Technology Landscape
Clinical trials run on software. The question is rarely whether a system is needed, it's which system does what, where the boundaries sit, and how the pieces connect.
What 21 CFR Part 11, Annex 11, and GAMP 5 Actually Mean for an SAP ICSM Implementation
Every SAP implementation in a pharmaceutical environment comes with a compliance layer. Everyone knows this. What is less often discussed is what that compliance layer actually requires in practice, and where it creates real implementation decisions rather than just documentation exercises.
